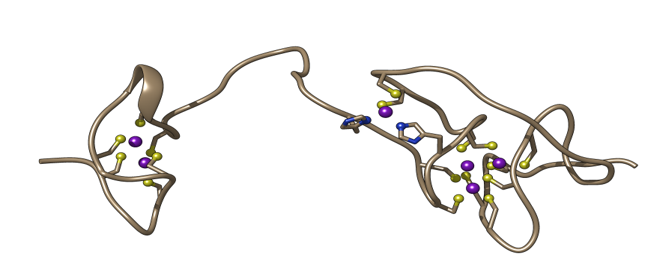
Role of Conserved Histidines in Zinc Selectivity and Structural Integrity of Sorghum Metallothionein SbMT4
SbMT4 functions as a zinc-specific metallothionein whose conserved histidine residues are essential for selective Zn binding, proper protein folding, and limiting cadmium incorporation, although altering these residues does not significantly change whole-plant metal accumulation, when expressed ectopically.
Keywords: Cadmium, Metalloprotein, Metallothionein, Micronutrients, Sorghum bicolor, Zinc
In this study, we demonstrated that histidine residues in the SbMT4 protein significantly reduce their relative affinity for cadmium ions compared to zinc ions, a finding confirmed both in vitro and in a cellular context. This demonstrates the potential of type 4 metallothioneins to selectively accumulate essential zinc while simultaneously limiting the storage of toxic cadmium in plant tissues. This is a crucial observation in the context of biofortification, as a plant-based diet is not only prone to result in zinc deficiency, but is also the major source of cadmium for non-smokers. – Mierek-Adamska
Zinc (Zn) and cadmium (Cd) share similar chemical properties, yet differ fundamentally in biological function: Zn is an essential micronutrient, whereas Cd is highly toxic, partly due to its ability to displace Zn in metalloproteins. Researchers from Nicolaus Copernicus University in Toruń and Warwick University investigated the role of sorghum type 4 metallothionein (SbMT4), a seed-specific protein proposed to act as a Zn-selectivity filter. Sequence comparisons revealed strong conservation of cysteine and histidine residues—key metal-binding ligands—across plant MT4 proteins. Recombinant expression and spectroscopic analyses demonstrated that wild-type SbMT4 predominantly forms a Zn₆ complex with a well-defined fold, confirming its classification as a Zn-thionein. Interestingly, Cd-expressed SbMT4 retained partial structural integrity due to the presence of a Zn ion in a mononuclear Cys₂His₂ site, highlighting the importance of histidine residues in stabilizing Zn binding and protein folding.
Mutational analyses replacing conserved histidines (His32 and His40) with tyrosine significantly altered metal-binding behavior and protein structure. These substitutions reduced Zn binding and increased Cd incorporation, with the double mutant (H32Y/H40Y) showing near-complete Cd preference and disrupted Zn coordination. Spectroscopic data revealed that all mutants exhibited misfolding of domain II regardless of the bound metal, underscoring the critical structural role of histidines in maintaining proper folding and metal selectivity. His40 was found to be particularly important for Zn/Cd discrimination. Despite these molecular effects, overexpression of wild-type and mutant SbMT4 in transgenic plants did not significantly alter total Zn or Cd accumulation in shoots, suggesting that SbMT4 primarily influences intracellular metal speciation rather than whole-plant metal content. Overall, the findings demonstrate that conserved histidines are essential for Zn-specific binding and structural integrity in SbMT4, with implications for engineering crops with improved micronutrient profiles and reduced heavy metal accumulation.
SorghumBase Examples:
SbMT4 (XP_002467575.1) has the locus ID SORBI_3001G311200. Type 4 plant metallothioneins (pMT4) are seed-specific proteins that may play a role in zinc binding. The Blindauer group investigated purified recombinant wild-type protein and site-directed mutants of key histidine residues using mass spectrometry, elemental analysis, and NMR spectroscopy. Their results demonstrated the importance of histidine residues and their critical role in metal selectivity.
However, overexpression of SbMT4 and its mutant proteins in Arabidopsis thaliana did not significantly affect the overall Zn and Cd concentrations in the shoots.
Reference:
Mierek-Adamska A, Gutierrez-Marcos J, Blindauer CA. Histidines promote zinc over cadmium binding to the single type 4 metallothionein from Great Millet (Sorghum bicolor). J Inorg Biochem. 2026 May;278:113227. PMID: 41570457. doi: 10.1016/j.jinorgbio.2026.113227. Read more
Related Project Websites:
- The Blinduer Lab webpage at University of Warwick: https://warwick.ac.uk/fac/sci/chemistry/research/blindauer/blindauergroup/
- The Gutierrez-Marcos Lab webpage at University of Warwick: https://warwick.ac.uk/fac/sci/lifesci/research/plantdevelopment/
- Agnieszka Mierek-Adamska’s contact details at Nicolaus Copernicus University in Toruń: https://www.bio.umk.pl/en/department-of-genetics/staff/